Aurora Biolabs Launches AI-Powered Antibody and Nanobody Design & Experimental Validation Services
Apr. 13, 2026
Aurora Biolabs is excited to announce the launch of its AI-driven antibody and nanobody design platform, combining state-of-the-art computational modeling with in-house experimental validation to accelerate biologics discovery.
Leveraging recent advances in protein structure prediction and generative AI, Aurora Biolabs integrates tools such as AlphaFold/AlphaFold2, Rosetta, and diffusion-based protein design models to enable the rational design of novel binders with enhanced affinity, specificity, and developability.
Technology Platform
Aurora Biolabs’ AI-enabled workflow incorporates:
Structure Prediction & Modeling
High-accuracy protein structure prediction using AlphaFold2 and related deep learning frameworks to model antigen–antibody interactionsGenerative Protein Design
Application of diffusion models and deep generative networks to design novel antibody and nanobody sequences targeting specific epitopesComputational Optimization
In silico affinity maturation and stability optimization using Rosetta-based energy calculations and machine learning-guided sequence refinementEpitope Mapping & Docking
Structure-based docking and interaction analysis to guide binder design and improve specificity
End-to-End Integrated Workflow
Aurora Biolabs uniquely combines AI design with experimental validation:
In Silico Design
Target analysis, epitope selection, and AI-driven antibody/nanobody generationGene Synthesis & Expression
Rapid production using mammalian, insect, or E. coli systemsPurification & Characterization
High-quality protein purification and QCExperimental Validation
Binding affinity (Biacor SPR analysis and Kd determination)
Functional assays
Biochemical and biophysical characterization
Bridging AI and Experimental Science
While AI-based protein design has rapidly advanced, experimental validation remains essential to confirm real-world performance. Aurora Biolabs addresses this critical gap by offering a closed-loop platform, enabling rapid iteration between computational design and laboratory validation.
This integrated approach significantly reduces development timelines and improves success rates in antibody discovery.
Applications
Therapeutic antibody discovery and optimization
Nanobody engineering for targeted delivery and diagnostics
Epitope-specific binder development
Diagnostic assay reagent development
Aurora Biolabs invites partners in biotechnology, diagnostics, and pharmaceutical development to leverage this next-generation AI-enabled platform to accelerate biologics innovation.
For more information, please contact:
📧 info@aurorabiolabs.com

Aurora Biolabs Signed A Collaboration Agreement with Structure Based Design, Inc., To Offer Co-Crystal Structure Determination Services to Customers.
Apr. 10, 2026
Structure Based Design, INC. or SBD, has been providing crystal structure and co-crystal structure determination services and actionable insights to both academic and industry clients since 2007. In almost two decades, it has partnered with over 70 biotech and pharma teams on more than 200 drug targets, contributing to 40+ IND (Investigational New Drug)-enabling candidates. Past collaborators include Zentalis, Quidel, Mirati, Effector, THPx, and academic groups such as Peter Schultz’s lab at Scripps and Dr. Kevan Shokat’s lab at UCSF.
Under the collaborative agreement, SBD now offers the same services to Aurora Biolabs customers at a fraction of the cost. Interested customers can schedule a free consultative meeting with SBD scientists by contacting Aurora Biolabs.


Aurora Biolabs Announces Offering of Assay Services with Customer-provided Compounds
Apr. 8, 2026
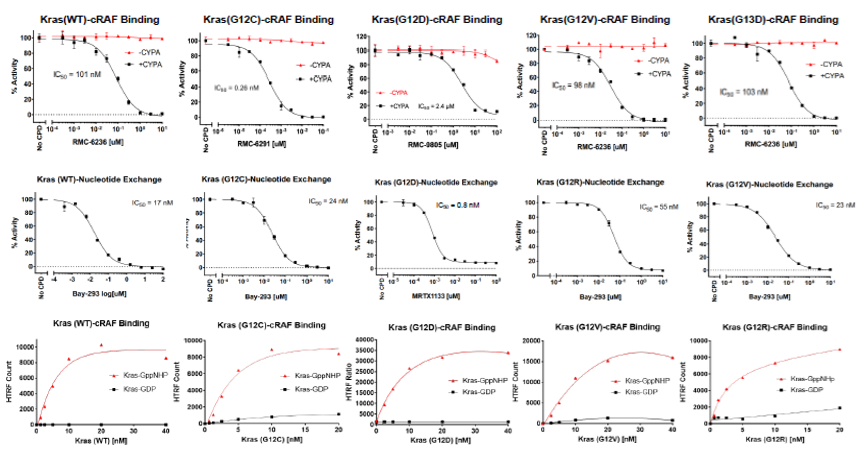
Aurora Biolabs, a premier developer and manufacturer of TR-FRET inhibitor screening assays in San Diego, CA, announces the offerings of assay services with all existing off-the-shelf in-house assays. These include popular Kras WT and mutant (G12C, G12D, G12V, G13D) TR-FRET inhibitor screening assays, CDK2/CyclinA2, CDK2/CyclinE1, CTLA-4/B-7, OX40/OX40L, PD-1/PD-L1, and DNA Polymerase Theta inhibitor screening assays.
Customers can provide compounds to Aurora Biolabs for IC50 determination per compound for just $200! High throughput (HTP) Inhibitor screening services are also available with customer-provided or -specified compounds or compound libraries, or with ~150,000 compounds from Aurora Biolabs in-house libraries. Prospective customers are encouraged to contact Aurora Biolabs to learn more about its assay services and how they can benefit drug discovery efforts.

Aurora Biolabs Launches Three New Assay Kits to Accelerate Kinase and Immunotherapy Drug Discovery
Jan. 28, 2026
Aurora Biolabs begins 2026 strong with the launch of three innovative assay kits designed to support high-throughput drug discovery: the CDK2/CyclinE1 Kinase Assay Kit, CDK2/CyclinA2 Kinase Assay Kit, and the CTLA-4:B7-1 Inhibitor Screening Assay Kit.
The CDK2/Cyclin E1 and CDK2/Cyclin A2 Kinase Assay Kits are no-wash, luminescence-based assays that utilize firefly luciferase to quantify residual ATP at the completion of kinase reactions. Optimized for 384-well plate formats, these assays are easily scalable for high-throughput screening applications, enabling efficient evaluation of kinase inhibitors.
The CTLA-4:B7-1 Inhibitor Screening Assay Kit is a no-wash TR-FRET–based assay, also designed for 384-well plates and readily adaptable to high-throughput inhibitor screening workflows. This assay supports the discovery and characterization of immune checkpoint modulators targeting the CTLA-4 pathway.
Together, these three assay kits provide powerful tools for researchers pursuing next-generation kinase therapeutics and immune-based drugs.
“We launched these three assay kits in January 2026 not because they are easy, but because they are important,” said COO May Zheng, MD at Aurora Biolabs.

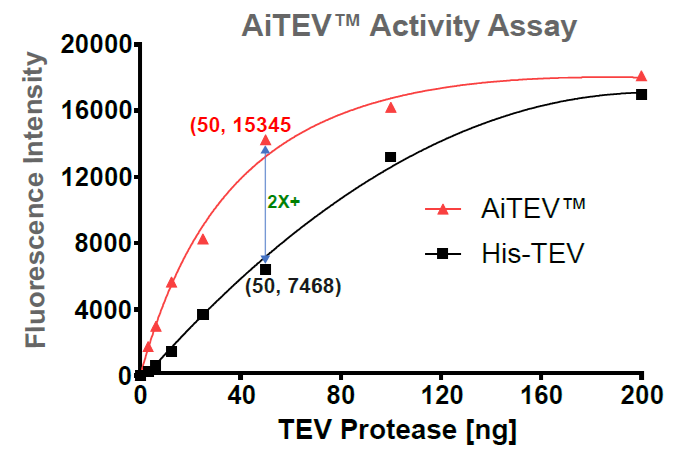
Aurora Biolabs Launches Artificially Engineered Tobacco Etch Virus Protease AiTEV™
Oct. 01, 2025
Aurora Biolabs today launched AiTEV™, an artificially engineered Tobacco Etch Virus protease. AiTEV™ is more active - at least 2 times more active than wild type (WT)/native Tobacco Etch Virus protease, highly site-specific, and significantly more stable than WT.
“This engineered version of Tobacco Etch Virus protease, AiTEV™, will bring significant advantages to large-scale biologics production, where purification tags are necessary to be removed post-purification,” says Dr. Kevin Duprez, Project Director of Aurora Biolabs.
For more information about AiTEV™, visit www.aurorabiolabs.com or contact sales@aurorabiolabs.com.
AiTEV™ Advantages:
-Highly Active
-Highly Site-Specific
-Highly Stable

Aurora Biolabs Launches $200 Rapid IC₅₀ and EC₅₀ Determination Service Using Proprietary TR-FRET Binding Assays and Activity Assays For Kras, PDL1, PARP, TEV, and More.
Aug. 18, 2025
Aurora Biolabs is pleased to announce a new analytical service for determining IC₅₀ (half-maximal inhibitory concentration) and EC₅₀ (half-maximal effective concentration) values for your compounds using our in-house developed KRAS (Product Page) and other TR-FRET assay kits.
Service Highlights:
Assay Platform: Aurora Biolabs’ validated KRAS assay kit with proven sensitivity and reproducibility.
Sample Requirement: 10 μL of compound at 10 mM concentration in DMSO or a dry powder.
Turnaround Time: Results delivered within 48 hours after sample receipt.
Pricing: $200 per duplicated assay — service fee only.
Output: Comprehensive report including raw data, assay curves, and calculated IC₅₀/EC₅₀ values.
With this service, customers can rapidly evaluate compound potency, compare analog series, and accelerate decision-making in drug discovery programs.
Contact Us today to discuss your project needs or request a quotation.
📧 info@aurorabiolabs.com
🌐 www.aurorabiolabs.com

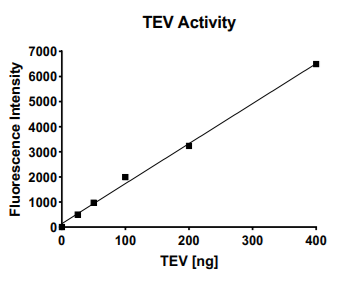
Aurora Biolabs Announces Official Launch of TEV Activity Assay kit
Jul. 30, 2025
TEV Activity Assay Kit has a wide range of applications. One of the popular uses is for optimizing cleavage conditions. Using Aurora Biolabs TEV Activity Assay Kit, factors that affect TEV activity, such as buffer composition, temperature, pH, reducing agents, or additives, can be tested and optimized. More applications can be found on the product brochure. Download a copy under the Resources tab.
The Aurora Biolabs Assay Development Team has more half a century of collective experience. Some have worked for companies that are well-recognized in the industry. All technical issues are addressed and resolved by the same scientists in a timely and effective manner. Contact sales@aurorabiolabs.com for a quote.
Assay Mechanism & Performance

Aurora Biolabs Launches TR-FRET PARP1 & PARP2 Inhibitor Screening Assays
Jul. 23, 2025
PARP1/PARP2 is often overexpressed in various cancers, including breast, ovarian, prostate, lung, and glioblastoma. This overexpression is thought to support tumor cell survival. Some PARP inhibitors not only block the catalytic activity of PARP1/PARP2 but also trap PARP1/PARP2 on DNA at sites of damage, preventing its release. This creates a toxic DNA-protein complex that interferes with DNA replication and repair, leading to cell death, particularly in cancer cells deficient in homologous recombination repair (e.g., BRCA1/2-mutant cells).
Asssay Mechanism & Performance
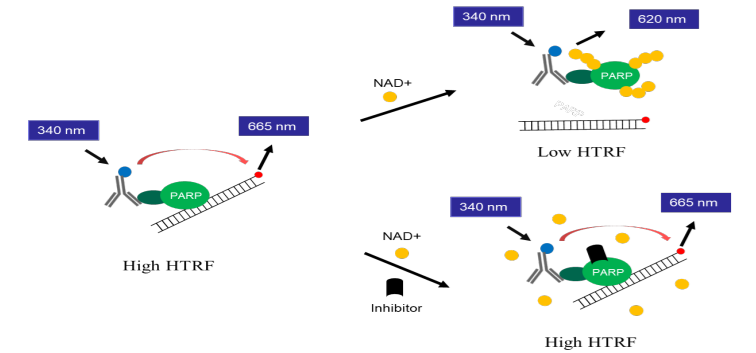
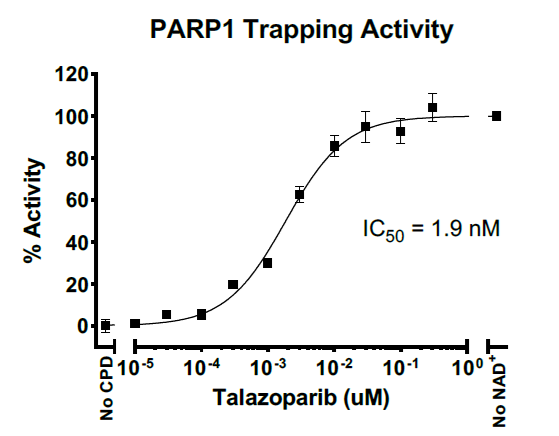
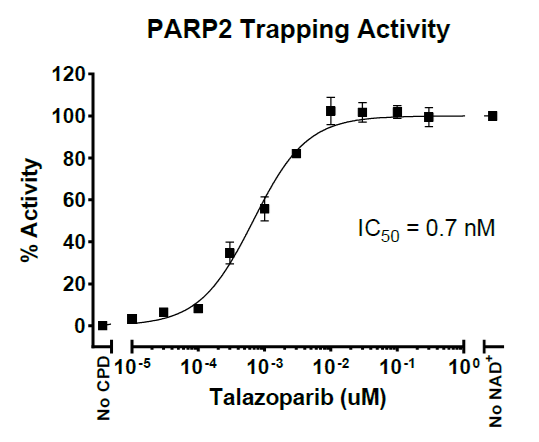

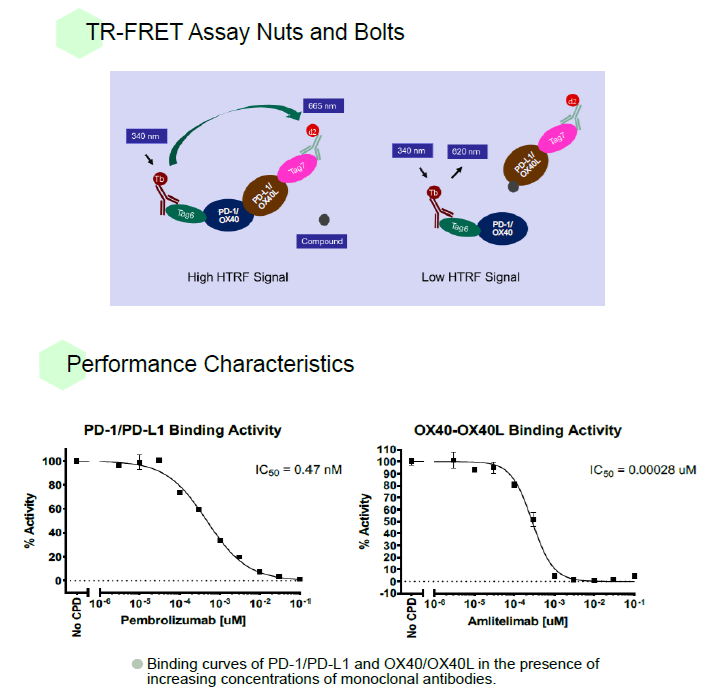
Aurora Biolabs Launches PD-1/PD-L1 and OX40/OX40L Inhibitor Screening Assays to Support Drug Discovery Efforts
May. 19, 2025
PD-1 and OX40 and corresponding ligands PD-L1 and OX40L, respectively, are attractive drug targets for immunotherapy. Aurora Biolabs has developed and manufactured TR-FRET assays for more than a decade. TR-FRET assays are know to be more sensitive than traditional enzyme-based assays due to the lanthanide-based donor fluorophores, which have long fluorescence lifetimes, and time-resolved detection, which minimizes background fluorescence interference.

Targeting the Oncogenic State of RAS with Tri-Complex Inhibitors: Aurora Biolabs Launches First Binding Assay for KRAS G12D/CypA/Inhibitor Screening
Mar. 28, 2025
Aurora Biolabs, a recognized leader in RAS-targeted protein production, assay development, and co-crystal structure determination, has announced the launch of the industry’s first binding assay for screening KRAS G12D/Cyclophilin A (CypA)/inhibitor tri-complexes. This milestone represents a significant leap in accelerating drug discovery for one of the most critical oncogenic targets in cancer biology.
KRAS mutations—particularly G12D—are frequently implicated in lung, colon, and pancreatic cancers, locking RAS proteins in a constitutively active GTP-bound "ON" state. These mutant proteins drive unchecked cell proliferation and tumor growth by persistently activating downstream effector pathways. While RAS has long been considered "undruggable," the emergence of tri-complex inhibitors offers a transformative approach. Revolution Medicines has pioneered this field with macrocyclic compounds that first bind Cyclophilin A, then selectively engage RAS(ON) to form a tri-complex that sterically blocks effector interaction.
To support the development of this next generation of RAS(ON) inhibitors, Aurora Biolabs now offers a proprietary binding assay capable of quantifying the formation of the KRAS G12D/CypA/inhibitor tri-complex. This assay delivers rapid, reliable data essential for screening compound libraries and optimizing drug candidates targeting the oncogenic RAS state. It complements Aurora's extensive toolkit, including high-quality KRAS mutant proteins, assay kits, and structure determination services, forming a complete solution for RAS-targeted drug discovery.
“Our goal is to deliver the fastest and most accurate tools to support KRAS-targeted therapy development,” said Dr. May Zheng, the project manager of Aurora Biolabs. “By enabling researchers to directly assess tri-complex formation in vitro, we are bridging a critical gap in translational oncology and accelerating the path toward effective therapies for KRAS-driven cancers.”
Aurora Biolabs invites collaboration with biotech and pharmaceutical partners aiming to explore or expand their RAS inhibitor pipelines. With the introduction of this cutting-edge assay, the company solidifies its position at the forefront of RAS drug discovery support.
For more information, visit www.aurorabiolabs.com or contact the Aurora Biolabs team at info@aurorabiolabs.com.

Aurora Biolabs Launches New Tag Removal Proteases
Oct. 07, 2024
Affinity tags are highly effective tools used for the expression and purification of recombinant proteins. However, these tags are not meant to be permanent fixtures on their respective recombinant proteins. Therefore, removal of an affinity tag is essential for further structural and functional studies of a specific protein.
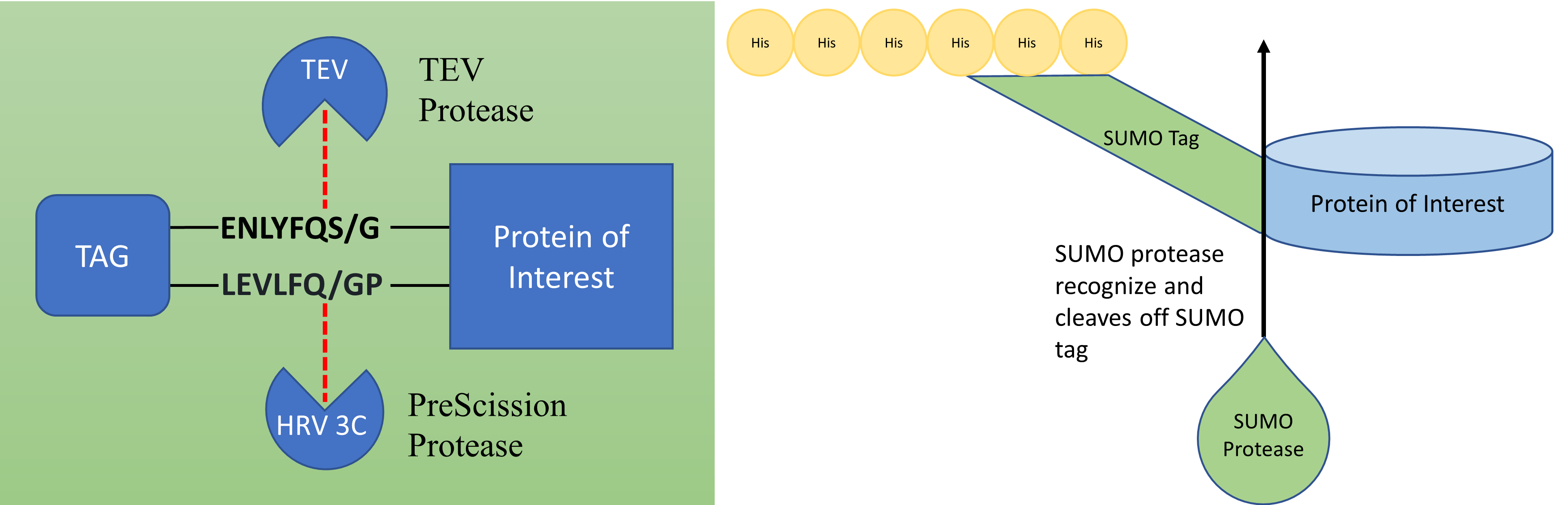
PreScission Protease (HRV 3C Protease) (Ready to use)
Recombinant SUMO Protease (ULP1) (Ready to use)
TEV Protease (Ready to use)

Aurora Biolabs Launches NEW SARS-CoV-2 Recombinant Proteins and Assay Kits to Support COVID-19 Research
Apr. 02, 2022
SARS-CoV-2, the viral agent responsible for the COVID-19 pandemic, is composed of an assortment of gene products critical to the viral lifecycle. We offer an assortment of SARS-CoV-2 recombinant proteins, kits, and services to advance your research and drug discovery needs.
SARS-CoV-2 Main/3CL Protease
SARS-CoV-2 Mpro Assay Kit
Recombinant SARS-CoV-2 Papain-like Protease
SARS-CoV-2 PLpro Assay Kit
Recombinant SARS-CoV-2 Helicase
Recombinant SARS-CoV-2 NSP7
Recombinant SARS-CoV-2 NSP8
About SARS-CoV-2 Protease assays:
Our protease activity assays for our kits/service are FRET based assays. The assay kits are designed to detect the cleavage of peptide substrates specific for the protease of interest, either Mpro or PLpro, and is designed for inhibitor screening applications. In general, the assay is fast and convenient, and requires just two steps. In the first step, the protease enzyme is preincubated with inhibitor for 30 minutes. The reaction is initiated by adding protease substrate at the second step. Fluorescence intensity is measured with a fluorescent plate reader at the excitation wavelengths of 340-360 nm and emission wavelengths of 460-480 nm.

Aurora Biolabs Launches New Human Recombinant Proteins and Assay Services for KRAS Research!
Mar. 01, 2022
The KRAS gene plays important roles in cell division, cell differentiation, and apoptosis. We offer an assortment of KRAS recombinant protein, kits, and services to advance your research and drug discovery needs.
Kras wild-type and mutants (G12C, G12D, G12R, G12V) (apo)
Kras wild-type and mutants (G12C, G12D, G12R, G12V) + GDP loaded
Kras wild-type and mutants (G12C, G12D, G12R, G12V) + GppNHp loaded (for activity binding assay)
Kras wild-type and mutants (G12C, G12D, G12R, G12V) TR-FRET based Nucleotide exchange assay kits
Kras wild-type and mutants (G12C, G12D, G12R, G12V) TR-FRET based Kras -cRAF binding assay
Kras assay services (Compound screening and profiling)
Human recombinant SOS1 and cRAF
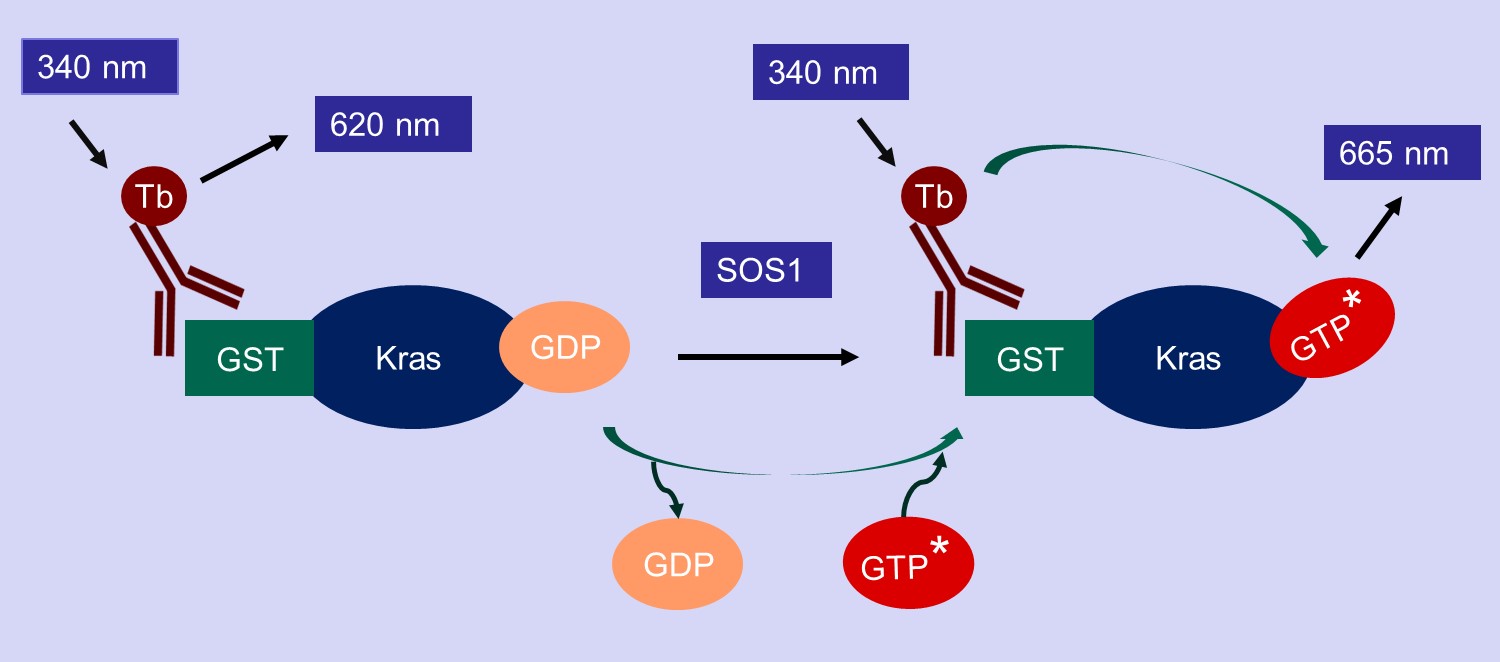
KRAS Nucleotide exchange assay
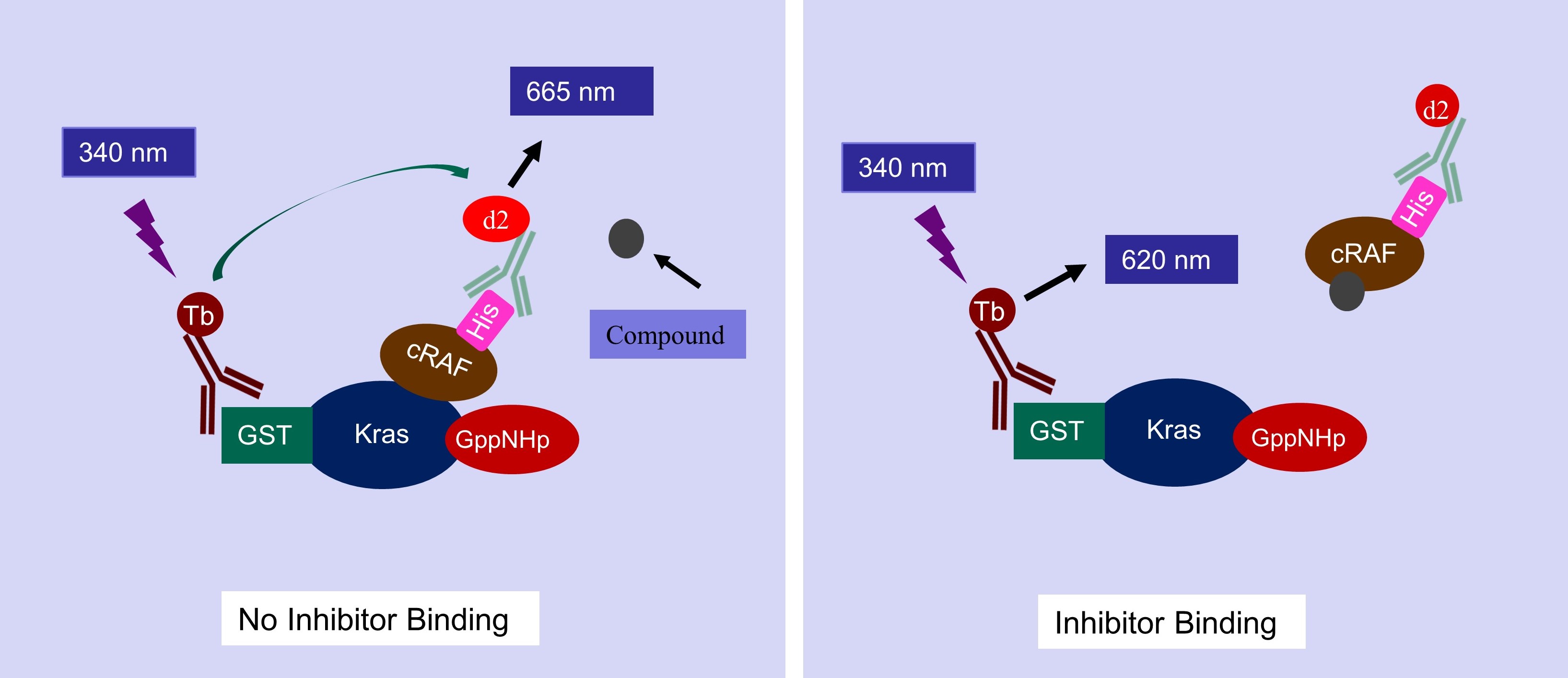
Our Nucleotide exchange assay for our kits/service is a TR-FRET based assay. The assay kit is designed to detect the GTP binding status of Kras in the presence of SOS1, a known Kras Guanine exchange factor. The Kras in our assay has a GST tag, that can bind to a Terbium-labeled anti-GST antibody (HTRF donor). If the Kras binds to a fluorescence-labeled GTP (HTRF acceptor), the donor and the acceptor will be brought in close proximity. Excitation of Terbium (340 nm) generates fluorescence resonance energy transfer (FRET) to fluorescence-labeled GTP acceptor, which emits specific fluorescence at 665 nm (figure below). Thus, GTP binding to the Kras can be quantitively measured by calculation of fluorescent ratio of 665 nm/620 nm.
Graphical Represntation of KRAS Nucleotide Exchange Assay Principle
Kras-cRAF binding assay
The Kras-cRAF binding assay is a TR-FRET based assay. In this assay, Kras is loaded with GppNHp, representing the activated Kras. The assay kit is designed to detect binding between Kras and cRAF. The Kras in this assay kit has a GST tag, that can bind to a Terbium-labeled anti-GST antibody (HTRF donor), and cRAF in this assay kit has a His tag, that can bind to a fluorescence-labeled anti-His antibody (HTRF acceptor). The binding of Kras with cRAF results in fluorescence resonance energy transfer (FRET) from the HTRF donor to the HTRF acceptor when the donor is activated allowing cRAF binding to be measured.
Graphical Represntation of Kras-cRAF Binding Assay Principle