Services at Aurora Biolabs
Assay Screening & Development Services
Scientists at Aurora Biolabs (an SBD company) and Structure Base Design, Inc. (SBD), have been providing assay development and assaying services to pharmaceutical/biotech companies and academia institutions for almost two decades. Our clients include Quidel, Mirati Therapeutics, Tanabe Research Laboratories, Caltech, and Salk Institute. We have consistently delivered quality results for our clients and partners. Many of our customers have submitted compounds discovered at Aurora Biolabs and SBD for clinical trials. The assays listed below, by no means exhaustive, are the assays that have been well-established at Aurora Biolabs. We also customize assays and develop de novo assays tailoring to each customer’s project and technical requirements.
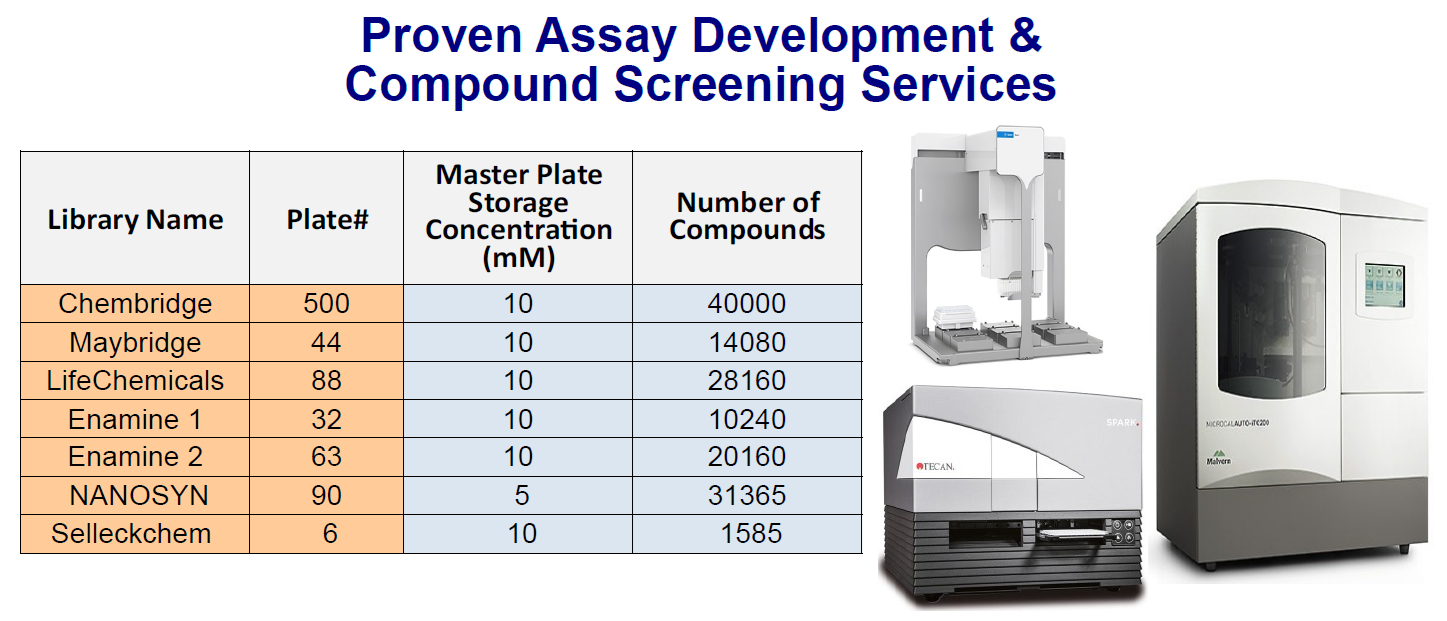
HTP Compound Library Screening With 145K Compounds
Structure-Based Drug Discovery (SBDD) is a well-established method for drug discovery. With the availability of increasingly powerful computers and ever-growing big data in the drug discovery space, SBDD is arguably the most efficient and powerful drug discovery process. Fragment-Based Drug Discovery (FBDD) evolved from the de novo drug design approach to further speed up the drug discovery process. Since the first Fragment-Based Drug Discovery (FBDD) drug, Zelboraf® (vemurafenib, PLX4032) was approved by FDA in 2011, there are now over 6 FDA-approved FBDD drugs on the market and 40+ FBDD drug candidates in clinical trials.
At Aurora Biolabs, an SBD company, we provide services in both SB-CADD (Structure Based-Computer Assisted Drug Discovery) screening and FBDD screening on more than 145,000 compounds. SBD can also provide crystallization service for drug target proteins and co-crystallization of drug target-compound. Contact Us for detail.
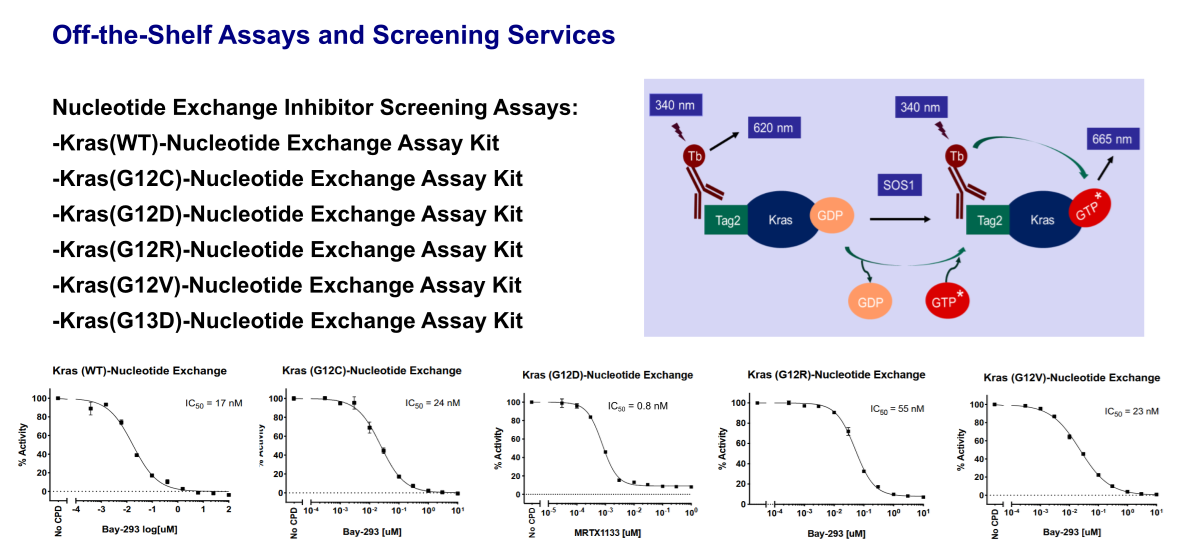
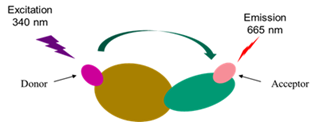
Kras Ligand Exchange Assay
Aurora Biolabs’ nucleotide exchange assay is a TR-FRET based assay. The assay kit is designed to detect the GTP binding status of Kras in the presence of SOS1, the most-studied guanine nucleotide exchange factor (GEF) of Kras. The Tag2-Kras in this assay kit is recognized by a Terbium-labeled anti-Tag2 antibody (HTRF donor). If Kras binds to a fluorescence-labeled GTP (HTRF acceptor), the donor and the acceptor will be brought in close proximity. Excitation of Terbium (340nm) generates fluorescence resonance energy transfer (FRET) to the fluorescence-labeled GTP acceptor, which consequently fluoresces at 665 nm (figure below). Thus, GTP binding to Kras can be quantitively measured by calculation of the fluorescent ratio of 665 nm/620 nm. The inhibitor blocking the nucleotide exchange will reduce the HTRF signal.
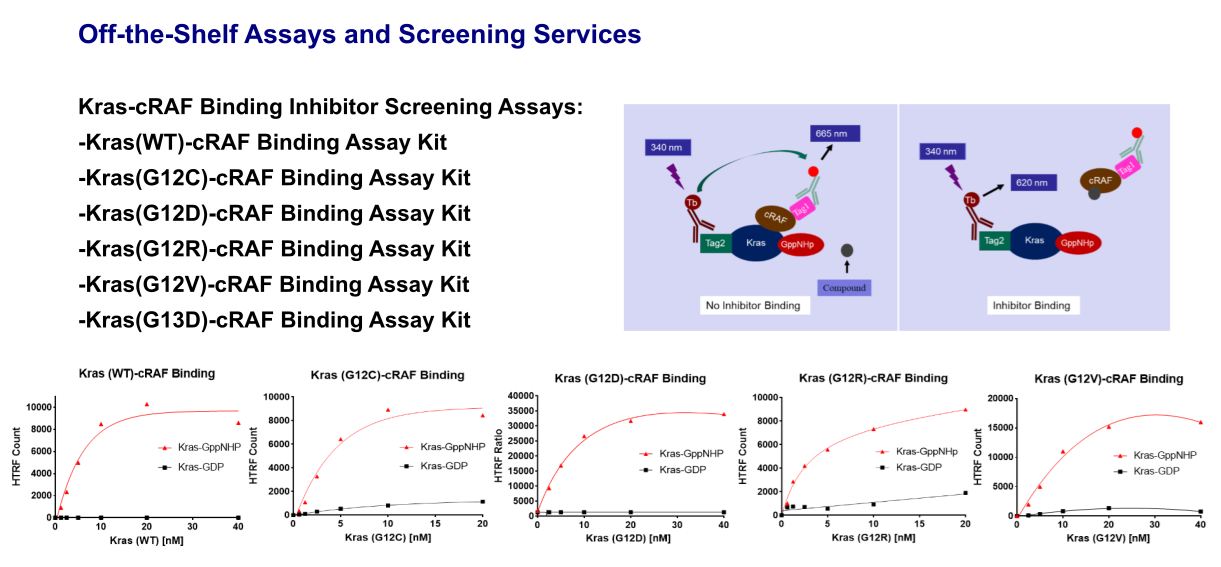
Kras-cRAF Binding Assay
The Kras-cRAF binding assay is a TR-FRET based assay. In this assay, Kras is loaded with GppNHp,representing the activated Kras. The assay kit is designed to detect binding between Kras and cRAF.
The Kras in this assay kit has a Tag2, that can bind to a Terbium-labeled anti-Tag2 antibody (HTRF donor), and cRAF in this assay kit has a Tag1, that can bind to a fluorescence-labeled anti-Tag1 antibody (HTRF acceptor). The binding of Kras with cRAF results in fluorescence resonance energy transfer (FRET) from the HTRF donor to the HTRF acceptor when the donor is activated allowing cRAF binding to be measured.
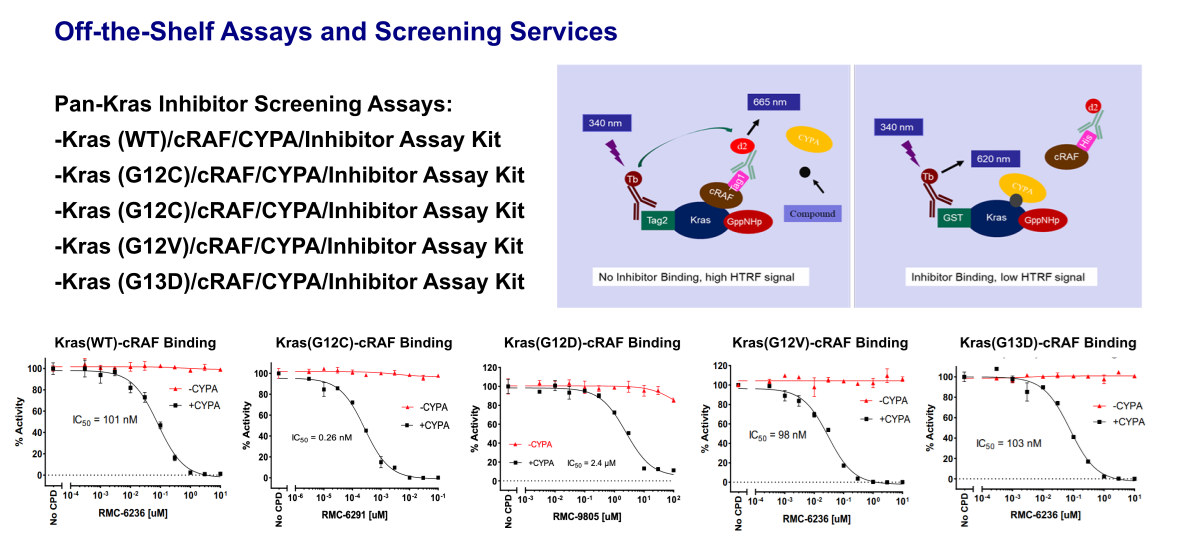
Kras-cRAF-CYPA Binding Assay
The Kras-cRAF-CYPA Inhibitor assay kit is a TR-FRET based assay, which is designed to screen Kras inhibitors and determine the Kras-inhibitor binding affinity. Tag2-Kras (WT) in this assay kit is loaded with GppNHp, which represents the activated Kras. The Ras binding domain (RBD) of cRAF in the kit has a Tag1 at N-terminus. A Terbium-labeled anti-Tag2 antibody binding to the Tag2-Kras serves as a fluorescence donor (HTRF donor), activation of which results in fluorescence resonance energy transfer (FRET) if Tag1-cRAF binds to the Kras, since the binding brings Terbium on the anti-Tag2 antibody close to the fluorophore on the anti-Tag1 antibody (HTRF acceptor). Thus, the binding status can be quantitively measured by calculating the ratio of the emission fluorescence intensity of the acceptor (665 nm) and donor (620 nm). If an inhibitor associated with CYPA binds to the Kras and blocks the cRAF binding, the HTRF signal will be reduced.
Other Inhibitors Screening Assays
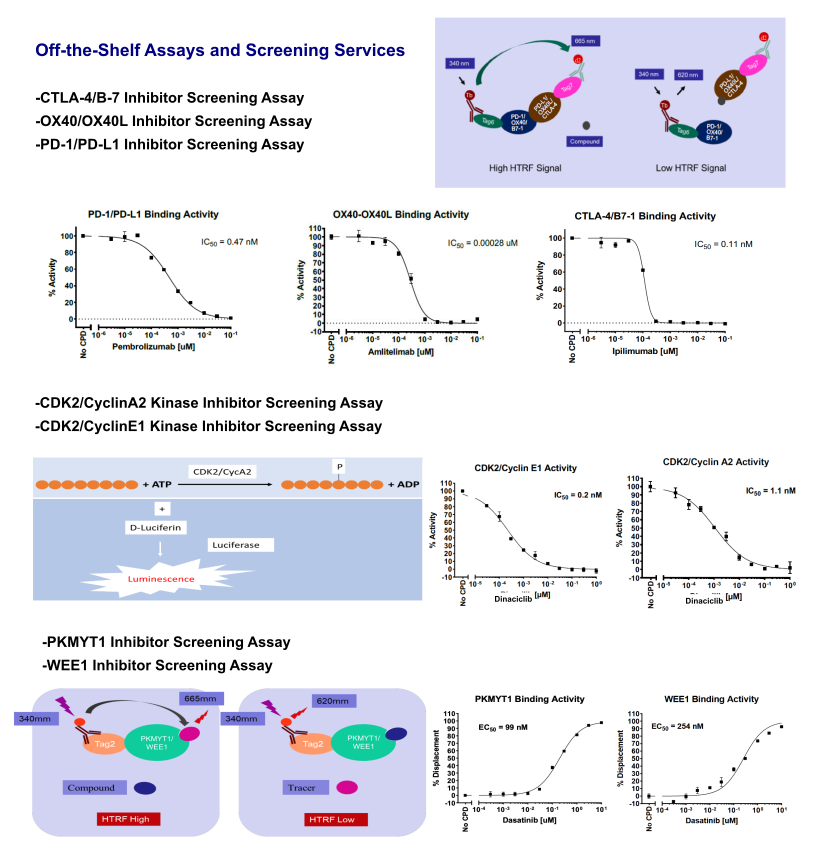
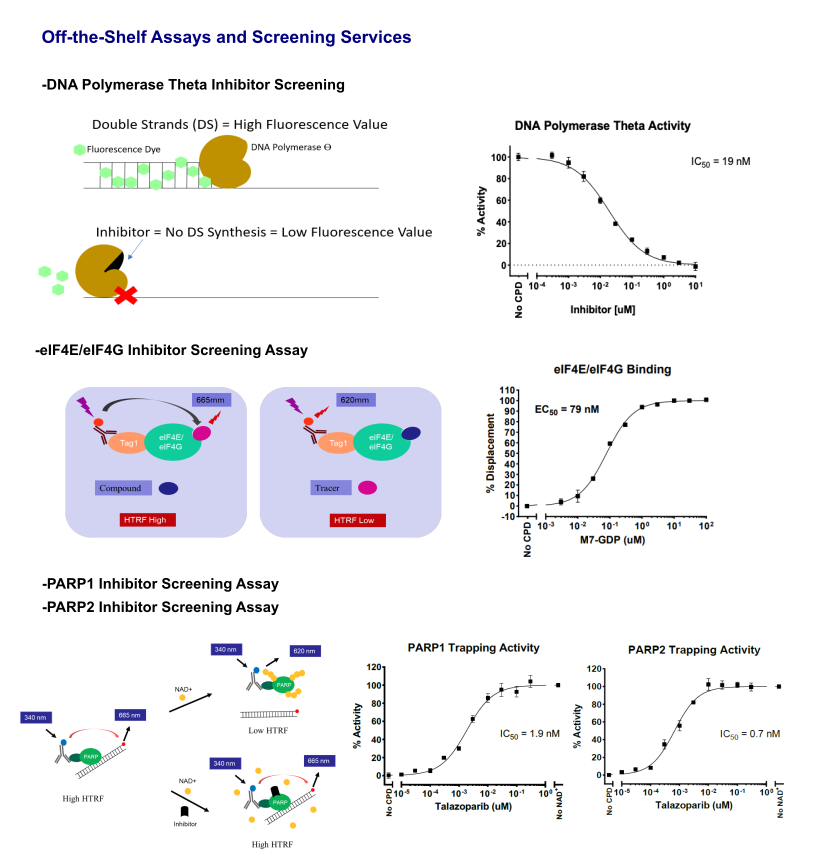
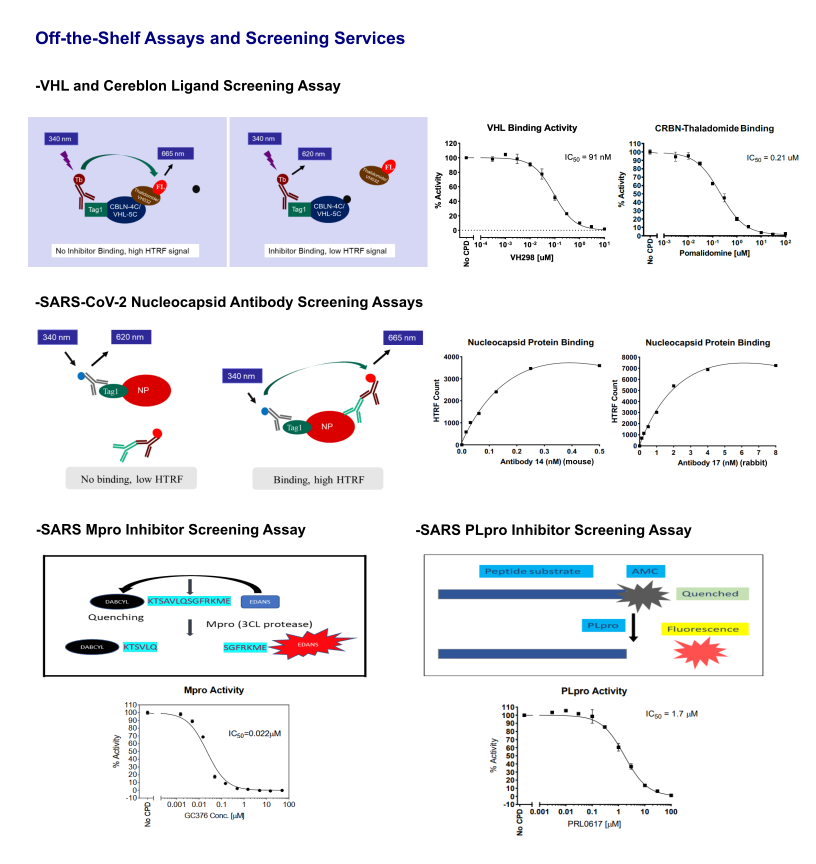
TR-FRET Custom Assay Development Services
A Time-Resolved Förster Resonance Energy Transfer (TR-FRET) assay is a biochemical technique that combines the principles of Time-Resolved Fluorescence (TRF) and Förster Resonance Energy Transfer (FRET) to achieve high sensitivity and reduced background noise. It is a powerful tool to study molecular interactions — such as protein–protein, receptor–ligand, or protein–nucleic acid binding — in a homogeneous (no-wash) format.
Principle of TR-FRET
FRET is a phenomenon where energy is transferred from a donor fluorophore to an acceptor fluorophore when they are within very close proximity (typically less than 10 nanometers). Two lanthanide complexes, europium and terbium, are commonly used donor fluorophores. When the donor is excited by light (LED or Laser) at 325 nm or 340 nm, it can transfer energy to the acceptor if they are within very close proximity (typically less than 10 nanometers), leading to acceptor fluorescence at a different wavelength (emission wavelength depending on the acceptor fluorophore used).
Advantages of TR-FRET Assay
Traditional FRET assays can suffer from high background fluorescence caused by interfering compounds and the short-lived emission from standard fluorophores. In the time-resolved florescence (TR-FRET) method, fluorescence is measured after a short delay (typically microseconds) following excitation, since lanthanides have a fluorescence lifetime that is much longer (microseconds to milliseconds) than the nanosecond-range fluorescence of background molecules. TR-FRET eliminates background fluorescence from biological samples and improves the signal-to-noise ratio.
TR-FRET assays are homogeneous high through-put assays with "mix-and-read" format, and do not require a physical separation or wash step to remove unbound labeled molecules, making them simple and efficient for automation.
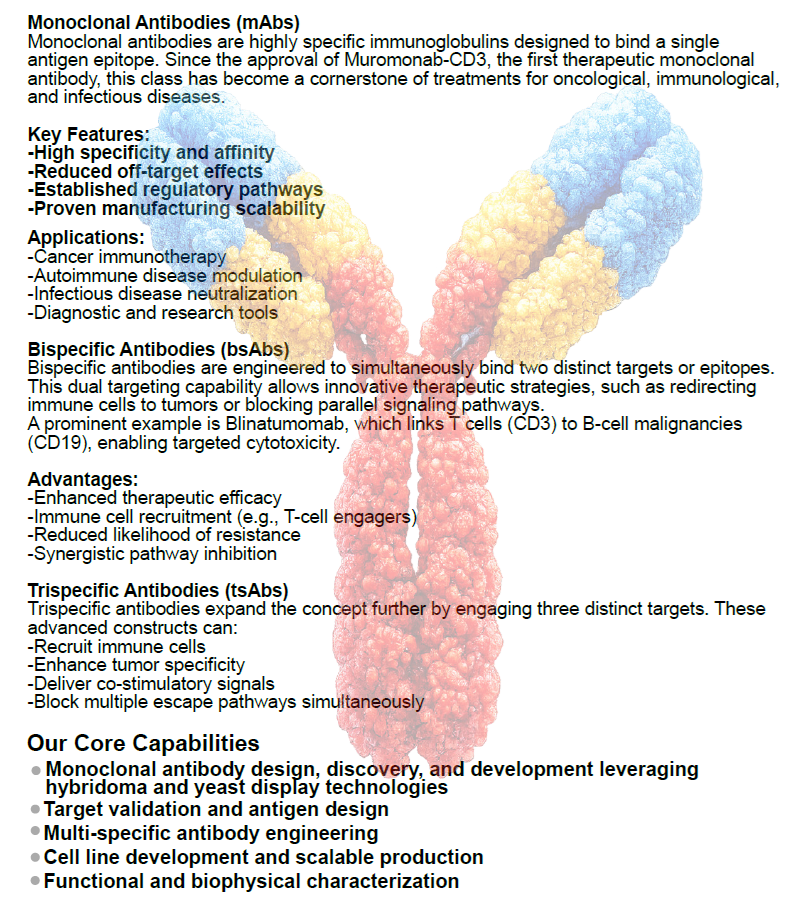
Antibody Development
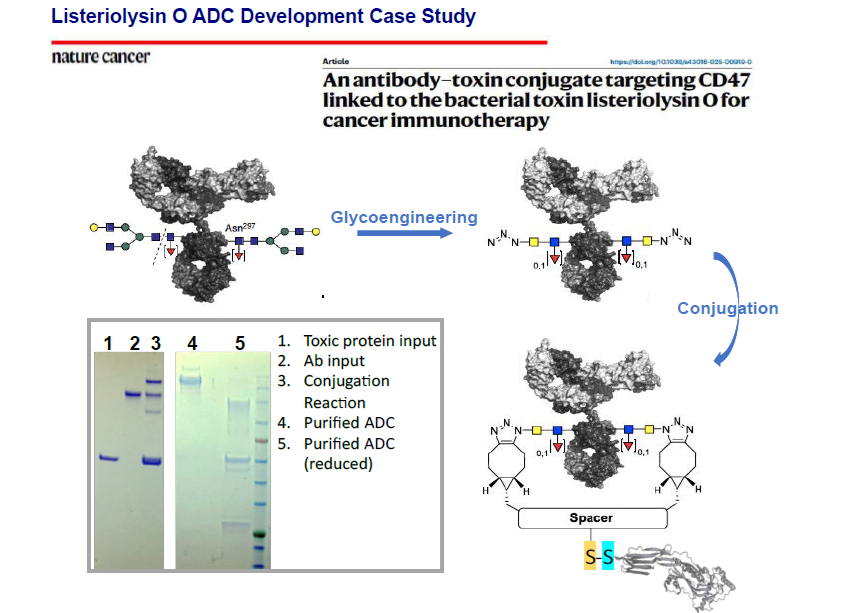
Antibody-Drug Conjugate Development

Protein Expression & Purification
Recombinant Protein production is a powerful technique used in the life sciences. The ability to express and purify a desired recombinant protein in large scale will allow scientists to explore a wide range of possibilities in protein research.
Aurora Biolabs services can take your project directly from gene synthesis to protein expression and recombinant protein purification. Protein products usually are purified with three different columns including affinity, ion exchange, and size-exclusion. The purity of the final products is greater than 95% and can be directly used for crystallization and other high-quality assays.
Specific activities and conformational homology of a protein are also major issues we consider during purification. We not only use general protecting reagents during the purification process, but also add various protection additives based on published literatures about the protein. Protein characterization of the final product can be tested in house or at the local Bioscience Service Center using Electrophoresis, Bioactivity study, Reversed-phase HPLC, Dynamic Light Scattering, Mass Spectroscopy Instruments, and BioCore.
Combining Fermenter, Akta Explorer, and large sized chromatography systems in the laboratory, we can produce quantities of protein samples ranging from 10 mgs to a few grams.